Breakthrough stent for aortic arch disease wins FDA approval, led by UChicago Medicine surgeon
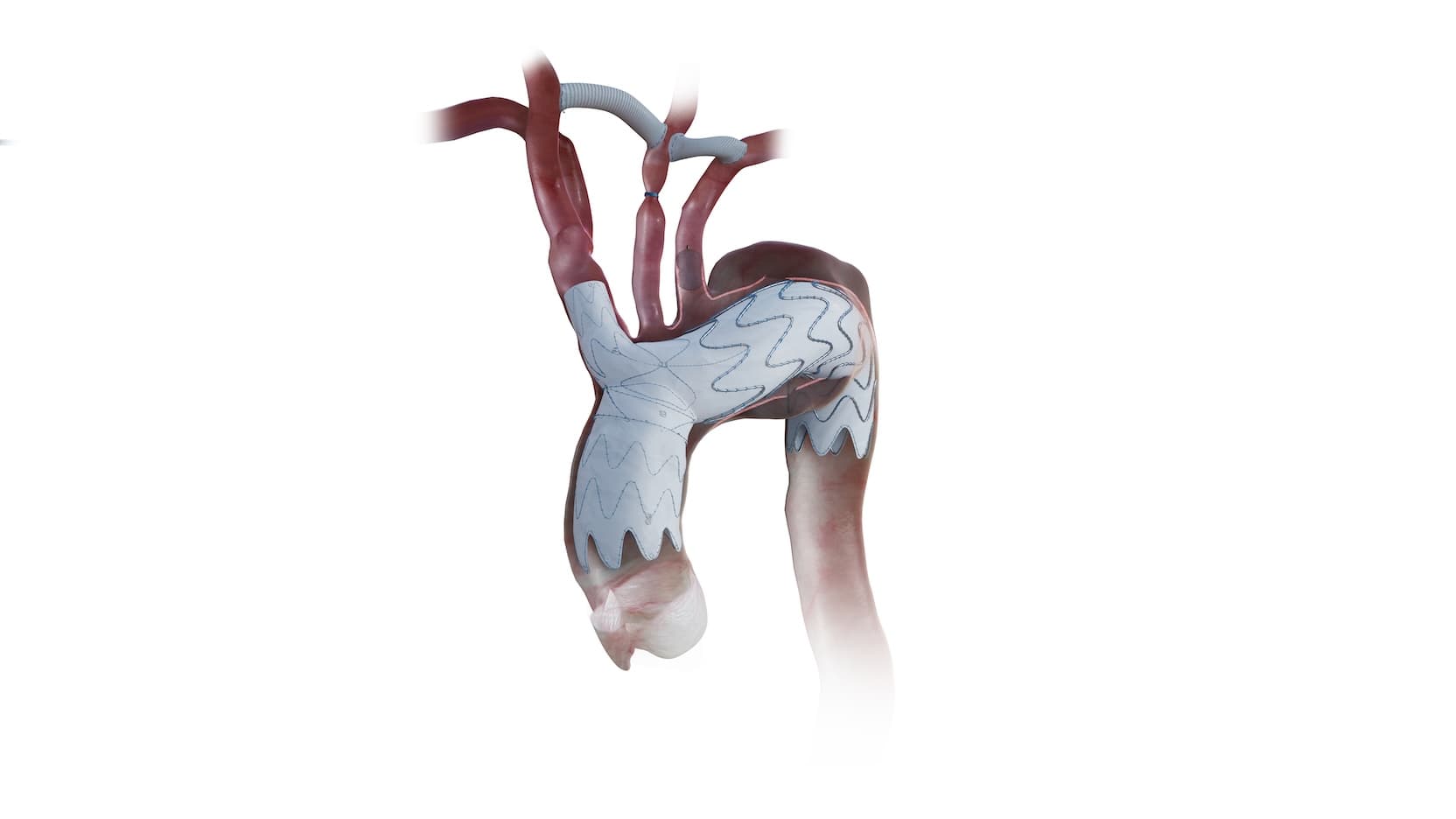
A first-of-its-kind stent to treat aortic arch disease in high-risk patients has been approved by the U.S. Food and Drug Administration, thanks to a clinical trial led by an internationally-recognized expert in vascular surgery at the University of Chicago Medicine.
One-year results from the TRIOMPHE clinical trial found the Nexus aortic arch stent graft to be effective in managing aortic arch disease, including chronic aortic dissections, in patients for whom open surgery is considered too dangerous.
The Nexus is the only FDA-approved stent with an extension piece intentionally designed for the ascending aorta, a notoriously difficult area for heart surgeons to treat.
“It’s a great demonstration of how physicians and industry can work well together, then work with the FDA to get the right clinical trial designed and get this out to patients who will truly benefit from a new device,” said Ross Milner, MD, Chief of Vascular Surgery and Endovascular Therapy at UChicago Medicine, and a co-principal national investigator for the TRIOMPHE study.
New option for aortic arch disease treatment
Aortic arch disease refers to disorders affecting the curved upper portion of the aorta that connects to the heart. Thoracic aneurysms, dissections and blockages are all types of aortic arch disease that can disrupt the normal flow of blood and cause life-threatening risks.
But many patients with aortic arch disease are considered too high risk or anatomically complex for surgery. Their options are limited to blood pressure medications and monitoring by their physician.
The Nexus stent offers a new option. It is a metal mesh tube inserted into the aorta to reinforce and repair the damaged or weakened section of the artery.
The stent is made of polyester fabric and nitinol — a memory metal that expands when heated to body temperature — and delivered through a small incision in the groin or right arm. Once in place, it acts like an internal sleeve that holds the artery open and restores normal blood flow.
“This gives us a less-invasive treatment option for patients who have had an aortic dissection repaired once, will likely need more dissection repair down the road and typically weren’t offered additional surgery because of the risk,” said Milner, who is also Senior Vice Chair of Perioperative Services and Clinical Excellence at UChicago Medicine.
“The device can also be used to treat aneurysms and type B dissections that have not had any prior surgery.”
Aortic stent study offers strong results
The prospective multicenter TRIOMPHE study, which is ongoing, is examining the safety and effectiveness of the stent in high-risk surgical patients with aortic arch disorders.
So far, results are encouraging: After one year, high-risk patients with chronic dissections who were treated with the Nexus stent showed safe and effective outcomes in the ascending aorta. Of the patients treated with the stent, 94% of them survived.
After 30 days, the device reduced serious complications, such as death and stroke, by 63% compared to the expected performance goal set by the FDA.
“One of the things we’re really proud of is the risk of stroke with this procedure is significantly lower than anything else that’s been evaluated,” Milner said.
The trial is led by Milner and co-principal national investigator Brad Leshnower, MD, director of thoracic aortic surgery at Emory Healthcare. The Nexus stent was developed by medical device company Endospan and approved in Europe in 2019.

Ross Milner, MD
Ross Milner, MD, is an internationally recognized expert in vascular surgery. He specializes in the treatment of complex aortic diseases.
Learn more about Dr. MilnerHeart and Vascular Center
From advanced diagnostic technology to minimally invasive treatments and robotic surgery, the University of Chicago Medicine can provide the full scope of care for any type of heart or vascular disorder in adults and children.
Explore our Heart and Vascular Center