Potential new therapy restores intestinal barrier for inflammatory bowel disease

Intestinal villi are small, finger-like projections that line the small instestine
In a new paper published online at Nature Medicine, a multi-institutional team of researchers describe how they identified a small molecule that can restore the intestinal barrier in disease and attenuate experimental immune-mediated colitis.
The researchers named this molecule ‘divertin’ because it diverts a key signaling molecule, MLCK, away from the intercellular junctions that regulate the intestinal barrier. The authors hypothesized that this, in turn, would prevent the immune system from disrupting the barrier and thus, interfere with a fundamental process required for inflammatory bowel disease (IBD) progression.
Barrier disruption is a critical component of multiple gastrointestinal diseases, including food allergy, celiac disease and IBD. It is also a key contributor to systemic diseases, including graft versus host disease, which can be a complication of bone marrow transplantation. Barrier restoration may be able to limit and even reverse these pathologies.
In this study, the researchers demonstrate that this novel approach can be an effective alternative to immunosuppressive therapies for acute and chronic intestinal disease. Their overall strategy of preventing signaling molecules from accessing particular intracellular sites also provides a new paradigm for safe and precise targeting of specific properties without globally disrupting signaling molecules that have multiple essential functions.
“This represents a completely novel, non-toxic approach to intestinal barrier restoration and treatment of inflammatory bowel disease,” said corresponding author Jerrold Turner, MD, PhD, (formerly at the University of Chicago Medicine) now at the Department of Pathology at Harvard Medical School.“Our study indicates that MLCK1 is a viable target for preserving epithelial barrier function in intestinal diseases and beyond,” he added. “This therapeutic approach may help break the cycle of inflammation that drives so many chronic diseases.”
A key fitting into a lock
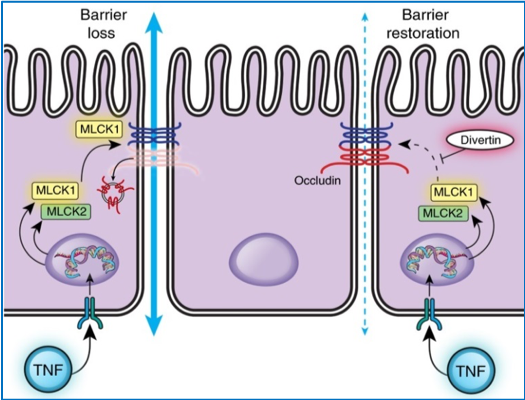
Discovering divertin wasn’t simple. The team queried a library of nearly 140,000 molecules to find one that fit into the signaling molecule, MLCK, like a key into a lock. They searched for attributes that would allow a drug to interrupt the process that creates tiny leaks, and thereby restore the barrier, without affecting other essential MLCK functions.
“We identify a domain-binding small molecule (divertin) that blocks MLCK1 recruitment without inhibiting enzymatic function,” they wrote. They soon found evidence that divertin could restore barrier function. It prevented MLCK1 from causing new injuries and shielded the mouse intestines from barrier loss and diarrhea. Similarly, in human intestinal tissue, there was “strong evidence that TNF-induced barrier loss was prevented or reversed.”
They also determined that divertin did not have significant toxicity. Daily intraperitoneal injections caused no serious problems. This allowed the authors to use divertin to treat mice with experimental IBD. They found that divertin effectively reduced severity and limited progression of this disease.
The authors note that these early studies suggest that this approach may be safe and effective at preventing or maintaining remission in IBD and in reducing severity of active disease. When compared to anti-TNF treatment — which has revolutionized care for IBD — divertin provided substantial benefits. These include limiting weight loss and reducing mortality.
“By these measures,” the authors note, “divertin was superior to anti-TNF treatment.” The combination therapy of divertin plus anti-TNF was even more effective at limiting disease.
Targeting barrier regulation with this strategy may also be useful in other diseases, such as celiac disease, atopic dermatitis, respiratory distress syndrome, graft versus host disease, and multiple sclerosis, the authors suggest. This approach, they add, “represents a new paradigm for enhancing specificity of small molecule therapies.”
“This new drug ameliorates inflammation and disease severity in mouse models of IBD and holds promise for patients with IBD as well as other diseases associated with barrier dysfunction,” according to a commentary by Julie Magarian Blander, PhD,Professor of Microbiology and Immunologyat Weill Cornell Medicine, in New York.
“Strikingly,” she added, “divertin outperformed anti-TNF in increased survival of treated mice, reduced fecal water content, immune cell infiltration and colonic histopathology, all of which are observed in patients with IBD.”
The 21-member research team was led by former University of Chicago Professor of Pathology Jerrold Turner. It also included lead author W. Vallen Graham (a University of Chicago graduate) and UCM Professor of Pathology Stephen Meredith.
Funding for this work was provided by the National Institutes of Health, the Broad Medical Research Foundation, the Department of Defense, a Catalyst Award from the Chicago Biomedical Consortium and the National Natural Science Foundation of China.
Inflammatory Bowel Disease Center
Outstanding patient care is at the center of everything we do. Our world-renowned specialists not only continue to push boundaries with their leading-edge research but also integrate the latest therapies into their treatment plans to offer you the most advanced care available.
Learn more about our IBD Center