Helmet-based ventilation is superior to face mask for patients with respiratory distress
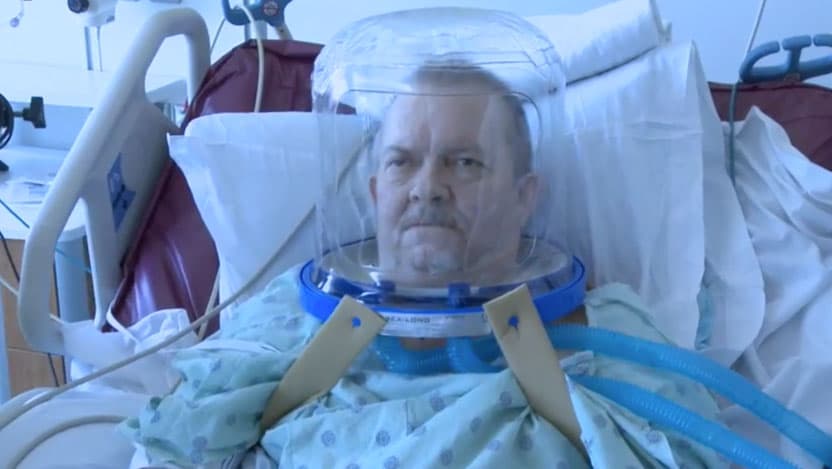
Striking news footage from Italy shows doctors treating coronavirus patients who are wearing bubble-shaped containers over their heads. These devices are used for patients suffering from acute respiratory distress syndrome (ARDS) caused by COVID-19, the disease caused by the 2019 novel coronavirus. The helmet surrounds the patient’s entire head to supply oxygen and is sealed with a soft, airtight collar that wraps around the neck.
In 2016, researchers from the University of Chicago Medicine led a study showing that using these helmets instead of standard face masks that cover the nose and mouth helps critically ill patients breathe better and can prevent them from needing intubation with a ventilator machine. Patients with helmet ventilation also spent less time in the intensive care unit and had better survival.
In a second study John P. Kress, MD, professor of medicine at the University of Chicago and senior author of the study. "The University's data and safety monitoring board recommended that we stop the trial early because the helmet consistently demonstrated multiple advantages, particularly the reduced need to intubate patients and longer-term reduction in mortality."
"After reviewing our data," he added, "the board felt that it would be difficult to justify enrolling more patients in the face mask arm of the trial, which exposed them to greater risks."
The helmet "confers several advantages over the face mask," the authors wrote. It is less likely to leak. This enables the care team to increase air pressure into the helmet, which helps keep the airway and lungs open and improves oxygen levels. It is also more comfortable, easier to tolerate because it doesn't touch the face, and patients can see through it well enough to watch television, talk or read.
Assembly and preparation of the helmet (video by Mark Saathoff)
Attach the ventilator's inspiratory and expiratory tubing to the port sites on the helmet. Set the ventilator to Pressure Support Mode. To improve patient ventilator synchrony, adjust the rise time to 50 milliseconds, and change the end flow to 50%. Stretch the rubber collar of the helmet and place over the patient's head.
Once the helmet is fully inflated, attach felt arms drops to the front and back of the helmet. This secures the helmet's position on the patient's head. The helmet pressurizes in seconds. PEEP is increased to achieve adequate lung recruitment and oxygen saturation.
Notice that at this level of PEEP there's no detectable leak in the ventilator circuit. FIO2 is also titrated to a non-toxic level to achieve oxygen saturations above 90%. Prior to application of the helmet, the patient was tachypneic and required 100% FIO2 to saturate at 93%. After transition to helmet non-invasive ventilation, the patient's vital signs have substantially improved. The patient is now saturating at 99% on pressure support of 5 with a PEEP of 15 and an FIO2 of 60%.
Patients who required the face mask for oxygenation for at least 8 hours were eligible to enroll in the study. Forty-four of the 83 patients who qualified to participate were then randomly assigned to the helmet group.
The other 39 were assigned to the face mask group.
All patients were severely ill with a 50 percent risk of requiring intubation or dying in the intensive care unit. About half of the patients had weakened immune systems from cancer or transplantation.
Patients in the helmet group, however, were three times less likely to require intubation, the study's primary endpoint. Only 18.2 percent of those wearing a helmet required an endotracheal tube, versus 61.5 percent of those wearing a face mask. The helmet group had, on average, more ventilator-free days (28 vs. 12.5).
Helmet patients also were more likely to survive. When compared at 90 days, 34 percent (15 patients) in the helmet group had died, compared to 56 percent (22 patients) in the face mask group.
Adverse trial-related events were minor. They included three skin ulcers for each group.
"These findings build on a shifting paradigm where less is more in the care of critically ill patients," said Bhakti Patel, MD, assistant professor of medicine and first author of the study. "We have chosen less sedation for more mental animation; less bed rest for more physical activity; and now we're choosing less intubations for more noninvasive ventilation."
The helmets used in this study are distributed by View a how-to video and access other helpful information about helmet-based ventilation.Helmet Ventilation Resources for Clinicians
